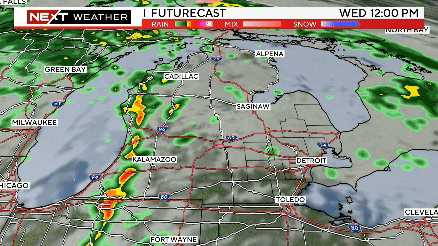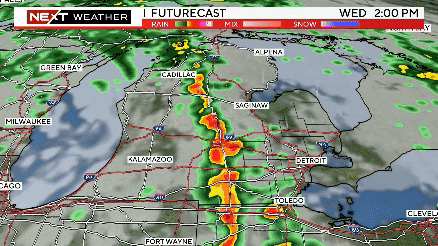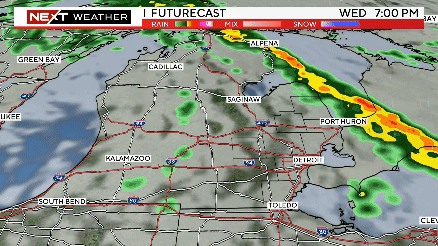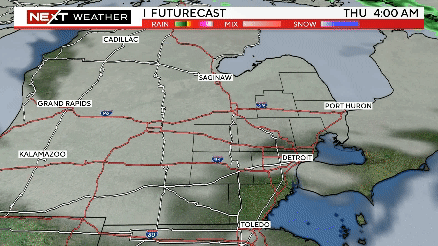
Severe weather rolls through Southeast Michigan on Wednesday
The timing of severe weather appears to be between 2-7 p.m., with the storm system moving through ahead of a cold front from west to east.
Watch CBS News

The timing of severe weather appears to be between 2-7 p.m., with the storm system moving through ahead of a cold front from west to east.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

America's mail carriers don't just face bad weather and aggressive dogs — they're also increasingly targets of violent crime.

Trillions of cicadas will emerge from the ground throughout the country this year, but only in a small area of Michigan.

A former school janitor in Michigan who is accused of posting photos of children in a social media group was recently indicted for possessing child pornography, officials said.

Michigan health experts are warning residents about the risks of consuming raw milk as the highly pathogenic avian influenza continues to affect dairy herds in the state.

Charli XCX and Troye Sivan are teaming up to co-headline an arena tour across North America, which will kick off here in Detroit.

The vehicles' failure to detect a "sudden degradation" in the battery could lead to to a sudden loss of power, Ford warns.

The Senate's 51-member Democratic majority voted to dismiss both charges as unconstitutional over the objections of Republican members.




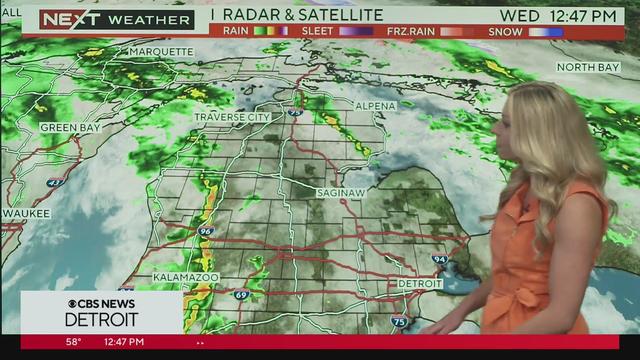
Severe storms possible this afternoon from 2pm until 7pm. Meteorologist Kylee Miller has the latest updates.

Walk for Miracles benefits programs of Beaumont Children's and takes place Saturday, June 8 at the Detroit Zoo in Royal Oak.

Just Between Friends is holding a community-building sales event where families can save hundreds of dollars. It is now located in more than 20 states.

Detroit Mayor Mike Duggan will deliver his eleventh State of the City; former Michigan House Speaker Lee Chatfield has been charged with embezzlement; Democrats regain control of the Michigan House after Tuesday's special elections and more on the latest top stories.
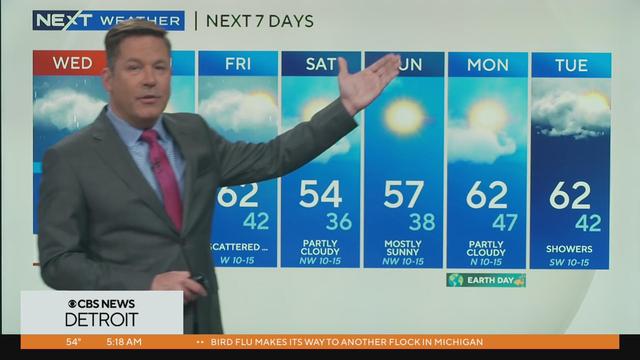
Starting with some rain and we roads. Storms roll in for the afternoon drive.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Charli XCX and Troye Sivan are teaming up to co-headline an arena tour across North America, which will kick off here in Detroit.

ABBA, Blondie and The Notorious B.I.G. are entering America's audio canon.

The annual Ford Fireworks show in Detroit is one of 20 shows nominated for USA Today's 2024 10Best Readers' Choice Places to See Fireworks contest.

More than 20 people have been convicted in the case, many of them onetime NBA players.

Gio Urshela had an RBI single and Matt Vierling scored on a wild pitch in the eighth inning to put Detroit ahead, and the Tigers' pitchers continued their solid play to beat the Texas Rangers 4-2.

Patrick Kane scored the shootout winner in a 5-4 Detroit win over the Montreal Canadiens in their regular-season finale Tuesday night, but the Red Wings were eliminated from playoff contention.

The penalties the university and the NCAA agreed on include three years of probation for the university, a fine and recruiting restrictions.

A whopping 10,000 athletes will carry the torch 3,100 miles over 68 days.

The timing of severe weather appears to be between 2-7 p.m., with the storm system moving through ahead of a cold front from west to east.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

America's mail carriers don't just face bad weather and aggressive dogs — they're also increasingly targets of violent crime.

Trillions of cicadas will emerge from the ground throughout the country this year, but only in a small area of Michigan.

A former school janitor in Michigan who is accused of posting photos of children in a social media group was recently indicted for possessing child pornography, officials said.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

General Motors and real estate firm Bedrock announced Monday the automaker's headquarters will move to the Hudson's Detroit development in downtown Detroit from the Renaissance Center.

With U.S. electric vehicle sales starting to slow, Ford Motor Co. says it will delay rolling out new electric pickup trucks and a new large electric SUV as it adds gas-electric hybrids to its model lineup.

New vehicle sales in the U.S. rose nearly 5% from January through March, as buyers stayed in the market despite high interest rates.

Reports claim that our vehicles may be collecting vast amounts of our personal data, information that could be shared with advertisers, data brokers, insurance companies and beyond.

Charli XCX and Troye Sivan are teaming up to co-headline an arena tour across North America, which will kick off here in Detroit.

ABBA, Blondie and The Notorious B.I.G. are entering America's audio canon.

The annual Ford Fireworks show in Detroit is one of 20 shows nominated for USA Today's 2024 10Best Readers' Choice Places to See Fireworks contest.

In the 1,000th episode, titled "A Thousand Yards," NCIS comes under attack by a mysterious enemy from the past.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

America's mail carriers don't just face bad weather and aggressive dogs — they're also increasingly targets of violent crime.

The vehicles' failure to detect a "sudden degradation" in the battery could lead to to a sudden loss of power, Ford warns.

The Senate's 51-member Democratic majority voted to dismiss both charges as unconstitutional over the objections of Republican members.

Elon Musk's 2018 compensation package is back for board re-certification after being voided by a Delaware court.

Gio Urshela had an RBI single and Matt Vierling scored on a wild pitch in the eighth inning to put Detroit ahead, and the Tigers' pitchers continued their solid play to beat the Texas Rangers 4-2.

MDARD officials say the virus, which was first detected in Michigan in 2022, was found in Newaygo County, making it the seventh flock in the state in the last two years.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.

State officials say the highly pathogenic avian influenza was detected in three dairy herds in Ionia, Isabella and Ottawa counties.

Most worrisome gaps involve cancer chemotherapy drugs, ER medications and and therapies for ADHD.

More than 20 people have been convicted in the case, many of them onetime NBA players.

Gio Urshela had an RBI single and Matt Vierling scored on a wild pitch in the eighth inning to put Detroit ahead, and the Tigers' pitchers continued their solid play to beat the Texas Rangers 4-2.

Patrick Kane scored the shootout winner in a 5-4 Detroit win over the Montreal Canadiens in their regular-season finale Tuesday night, but the Red Wings were eliminated from playoff contention.

The penalties the university and the NCAA agreed on include three years of probation for the university, a fine and recruiting restrictions.

A whopping 10,000 athletes will carry the torch 3,100 miles over 68 days.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

Riney, who has spent his 46-year career at Henry Ford Health, was named one of Modern Healthcare's 100 Most Influential People.

Severe storms possible this afternoon from 2pm until 7pm. Meteorologist Kylee Miller has the latest updates.

Walk for Miracles benefits programs of Beaumont Children's and takes place Saturday, June 8 at the Detroit Zoo in Royal Oak.

Just Between Friends is holding a community-building sales event where families can save hundreds of dollars. It is now located in more than 20 states.

Detroit Mayor Mike Duggan will deliver his eleventh State of the City; former Michigan House Speaker Lee Chatfield has been charged with embezzlement; Democrats regain control of the Michigan House after Tuesday's special elections and more on the latest top stories.

Starting with some rain and we roads. Storms roll in for the afternoon drive.