
Woman files sexual harassment lawsuit against Metro Detroit chiropractor
Attorney Azzam Elder filed a lawsuit against John Pispidikis and Spinal Recovery Center in Warren, alleging sexual harassment.
Watch CBS News

Attorney Azzam Elder filed a lawsuit against John Pispidikis and Spinal Recovery Center in Warren, alleging sexual harassment.

Prosecutors charged James Freeman, 41, and Christopher Hash, 42, after drugs and a handgun were seized.

In December 2023, Detroit Wing Company gifted its Roseville location to Rising Stars Academy, a school for young adults with intellectual and developmental disabilities.

The Macomb County Prosecutor's Office announced new charges have been filed against former police officers Dammeon Player and Carlos Taylor, accused of using excessive force in 2023.

It was an eventful week in Lansing for government transparency, as Michigan's Attorney General and Secretary of State both testified in favor of a package of bills that would increase government transparency and accountability.

After he became gravely ill, an Oakland County man from Highland, Michigan, braced for the ultimate fight to survive, is sharing the remarkable story of his recovery.

UAW claims historic victory, with an overwhelming majority of VW workers at Chattanooga factory voting to unionize.

Warren police are investigating whether the gun was stored properly.

Police are searching for Joi Boswell, who was last seen on March 15 leaving with a man in a red vehicle.





A Metro Detroit chiropractor and his place of employment are at the center of a sexual harassment lawsuit. Attorney Azzam Elder said he filed a lawsuit Friday against John Pispidikis and Spinal Recovery Services on behalf of his client, a former patient.

In December 2023, Detroit Wing Company gifted its Roseville location to Rising Stars Academy, a school for young adults with intellectual and developmental disabilities.

Detroit City FC returns to USL Championship action this Saturday at Keyworth Stadium as they prepare to host Oakland Roots SC.

Democratic lawmakers say they're seeing traction on these bills because they have control of both chambers in the legislature.
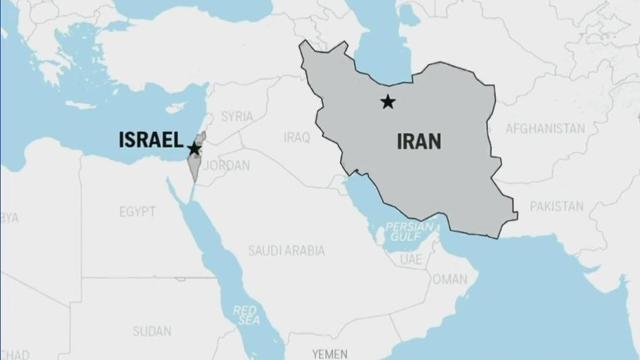
Javed Ali, a University of Michigan professor and national security expert, breaks down Israel's retaliation against Iran for an attack.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.
Michigan State is breaking in a new football coach and quarterback this year.

The Wolverines had a record 18 players at the combine, giving them a chance to be the first school with at least 16 prospects picked in one NFL draft.

Sherrone Moore will have a lot of big decisions to make in his first season as Michigan's coach, leading the defending national champions after Jim Harbaugh bolted to coach the Los Angeles Chargers.

The NFL draft is coming to Detroit and for a change, the Lions are coming off a successful season and have appeared to address every need with seemingly sound moves.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.

Attorney Azzam Elder filed a lawsuit against John Pispidikis and Spinal Recovery Center in Warren, alleging sexual harassment.

Prosecutors charged James Freeman, 41, and Christopher Hash, 42, after drugs and a handgun were seized.

In December 2023, Detroit Wing Company gifted its Roseville location to Rising Stars Academy, a school for young adults with intellectual and developmental disabilities.

The Macomb County Prosecutor's Office announced new charges have been filed against former police officers Dammeon Player and Carlos Taylor, accused of using excessive force in 2023.

It was an eventful week in Lansing for government transparency, as Michigan's Attorney General and Secretary of State both testified in favor of a package of bills that would increase government transparency and accountability.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

General Motors and real estate firm Bedrock announced Monday the automaker's headquarters will move to the Hudson's Detroit development in downtown Detroit from the Renaissance Center.

With U.S. electric vehicle sales starting to slow, Ford Motor Co. says it will delay rolling out new electric pickup trucks and a new large electric SUV as it adds gas-electric hybrids to its model lineup.

New vehicle sales in the U.S. rose nearly 5% from January through March, as buyers stayed in the market despite high interest rates.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.

Charli XCX and Troye Sivan are teaming up to co-headline an arena tour across North America, which will kick off here in Detroit.

ABBA, Blondie and The Notorious B.I.G. are entering America's audio canon.

UAW claims historic victory, with an overwhelming majority of VW workers at Chattanooga factory voting to unionize.

A person self-immolated at a park across from the courthouse where former President Donald Trump's New York criminal trial is taking place.

The Wolverines had a record 18 players at the combine, giving them a chance to be the first school with at least 16 prospects picked in one NFL draft.

The MY 2024 Cybertrucks have faulty accelerator pedals that may be dislodged when high force is applied, the company said.

Starbucks unveiled the new cups ahead of Earth Day and as a new report warns plastic production emissions are even greater than those from aviation.

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

MDARD officials say the virus, which was first detected in Michigan in 2022, was found in Newaygo County, making it the seventh flock in the state in the last two years.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.
Michigan State is breaking in a new football coach and quarterback this year.

The Wolverines had a record 18 players at the combine, giving them a chance to be the first school with at least 16 prospects picked in one NFL draft.

Sherrone Moore will have a lot of big decisions to make in his first season as Michigan's coach, leading the defending national champions after Jim Harbaugh bolted to coach the Los Angeles Chargers.

The NFL draft is coming to Detroit and for a change, the Lions are coming off a successful season and have appeared to address every need with seemingly sound moves.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

A Metro Detroit chiropractor and his place of employment are at the center of a sexual harassment lawsuit. Attorney Azzam Elder said he filed a lawsuit Friday against John Pispidikis and Spinal Recovery Services on behalf of his client, a former patient.

In December 2023, Detroit Wing Company gifted its Roseville location to Rising Stars Academy, a school for young adults with intellectual and developmental disabilities.

Detroit City FC returns to USL Championship action this Saturday at Keyworth Stadium as they prepare to host Oakland Roots SC.

Democratic lawmakers say they're seeing traction on these bills because they have control of both chambers in the legislature.

Javed Ali, a University of Michigan professor and national security expert, breaks down Israel's retaliation against Iran for an attack.