
Ford's first-quarter net income falls 24% as combustion engine unit sees sales, revenue decline
Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.
Watch CBS News

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

One of the NFL's biggest events begins Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

The Supreme Court will convene Thursday to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

De La Salle Collegiate High School senior Kandakai Sherman will be handing NFL prospects their hat for the team they've been selected by.

Here's a list of some things to do in the Motor City while you're in town for the NFL Draft.

CDC's provisional figures show a 2% decline in births from 2022 to 2023.

As thousands enter downtown Detroit, you can expect long lines and not much space, especially parking.

Detroit is just hours away from hosting the 2024 NFL Draft. Celebrities and football stars are making their way into town, along with hundreds of thousands of visitors, and Detroit business wonders are ready for them.




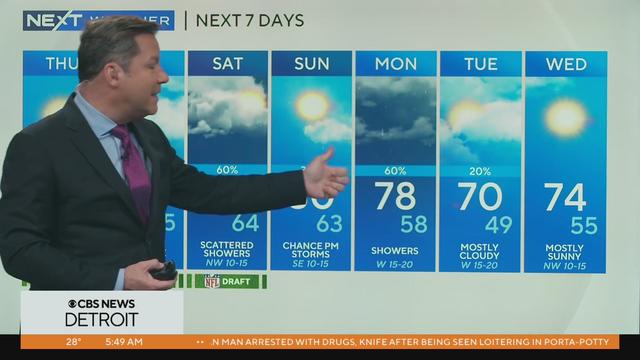
Warming near 80 this weekend.

De La Salle Collegiate High School senior Kandakai Sherman will be handing NFL prospects their hat for the team they've been selected by.

The 2024 NFL Draft preparations reach the 11th hour. Now the city braces for one of the largest sporting events. As thousands enter downtown Detroit, you can expect long lines and not much space, especially parking.
Detroit is just hours away from hosting the 2024 NFL Draft. Celebrities and football stars are making their way into town, along with hundreds of thousands of visitors, and Detroit business wonders are ready for them.

As the excitement ramps up, we're taking a look at the business of the NFL Draft in Detroit. We sat down with Michigan. Gov. Gretchen Whitmer, Detroit Mayor Mike Duggan, Wayne County Executive Warren Evans, Mark Lee and sportscaster Jeff Lesson to talk about one of the biggest events in the NFL.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

One of the NFL's biggest events begins Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

The 5,400-square-foot Lions fan experience at Hart Plaza is bringing Ford Field to the heart of the NFL footprint to get fans pumped for the draft as well as the upcoming football season.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

Here's what you should do if your child gets lost while your at the NFL Draft this weekend.

Here's a list of some things to do in the Motor City while you're in town for the NFL Draft.

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

One of the NFL's biggest events begins Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

The Supreme Court will convene Thursday to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

De La Salle Collegiate High School senior Kandakai Sherman will be handing NFL prospects their hat for the team they've been selected by.

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Despite a small dip in U.S. vehicle sales, General Motors' first-quarter net income rose more than 25% on strong deliveries of pickup trucks and other higher-profit vehicles.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

The Supreme Court will convene Thursday to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

CDC's provisional figures show a 2% decline in births from 2022 to 2023.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.

This 2024 "State of the Air" report warns efforts to reduce emissions are undermined by extreme heat, drought and wildfires caused by climate change.
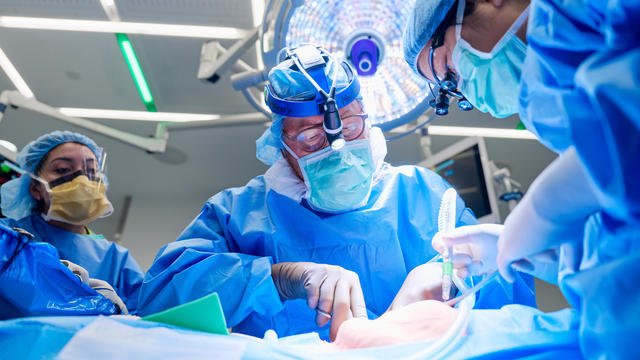
For the first time, surgeons at NYU Langone Health performed a combined mechanical heart pump and gene-edited pig kidney transplant into a living person.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.
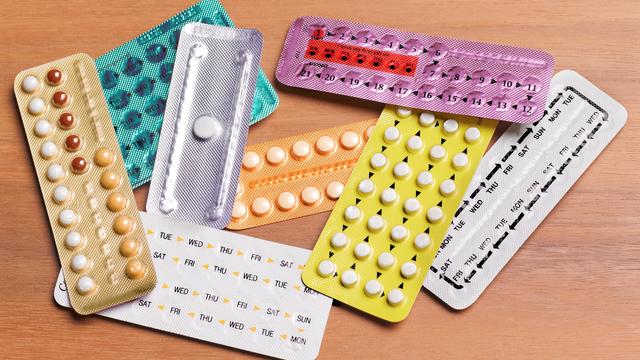
The Michigan Legislature is considering two bills that could make getting access to hormonal birth control a little easier.

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

One of the NFL's biggest events begins Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

The 5,400-square-foot Lions fan experience at Hart Plaza is bringing Ford Field to the heart of the NFL footprint to get fans pumped for the draft as well as the upcoming football season.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

Here's what you should do if your child gets lost while your at the NFL Draft this weekend.

Here's a list of some things to do in the Motor City while you're in town for the NFL Draft.
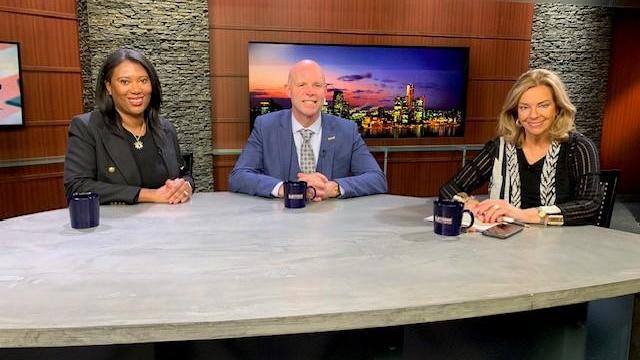
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

Warming near 80 this weekend.

De La Salle Collegiate High School senior Kandakai Sherman will be handing NFL prospects their hat for the team they've been selected by.

The 2024 NFL Draft preparations reach the 11th hour. Now the city braces for one of the largest sporting events. As thousands enter downtown Detroit, you can expect long lines and not much space, especially parking.

Detroit is just hours away from hosting the 2024 NFL Draft. Celebrities and football stars are making their way into town, along with hundreds of thousands of visitors, and Detroit business wonders are ready for them.

As the excitement ramps up, we're taking a look at the business of the NFL Draft in Detroit. We sat down with Michigan. Gov. Gretchen Whitmer, Detroit Mayor Mike Duggan, Wayne County Executive Warren Evans, Mark Lee and sportscaster Jeff Lesson to talk about one of the biggest events in the NFL.