
Detroit Lions unveil 2024 modern uniforms, including black alternate
Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.
Watch CBS News

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.

Former Michigan Police Chief Tressa Beltran has pleaded guilty to drug charges, according to the Michigan Attorney General's office.

Two U.S. officials confirm to CBS News that an Israeli missile has hit Iran, in apparent retaliation for last weekend's drone and missile attack on the Jewish state.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

In response, one House Democrat said she is surprised to continue having these conversations.

The Detroit Zoo is planning to reveal a new design for its water tower this year.

A Detroit man was taken into custody after police said he went to a 7-11 store in Livingston County to meet who he thought was a 14-year-old girl.

The rent is high not just in Detroit, where, according to nonprofit Higher Ground Abodes, the city has just a quarter of the affordable homes needed, but also across the state.

The city of Detroit on Thursday announced the fourth phase of closures, which will begin at 6 a.m. on Saturday, April 20, through 11:59 p.m. on Tuesday, April 30.





Iraqi Prime Minister Mohammed Shia al-Sudani traveled to Michigan on Thursday to meet with the state's large Iraqi community and update them on escalating tensions in the Middle East.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform.

In celebration of Arab American Heritage Month, CBS News Detroit takes a look into the culture in Metro Detroit, highlighting the history, leadership, food, and comedy.
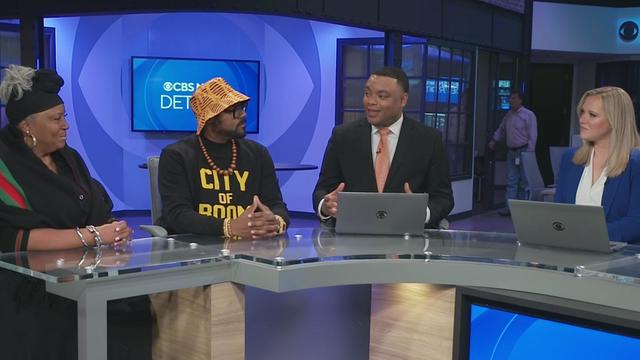
Behind every athlete is a mother supporting them behind the scenes. On April 21, the "My Mom's First Fan" event celebrates Metro Detroit football mothers. Khary Frazier, founder of Detroit is Different, and Constance "Maspice" Davis, the mother of retired NFL player Anthony "Spice" Adams, joined CBS News Detroit to talk about the upcoming event.

Rain overnight will break early tomorrow morning while cooler air rushes in to kickstart our weekend.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Taylor Swift took to social media hours ahead of the expected release of her new album "The Tortured Poets Department."

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.

On the ground, the Michigan State Police will send at least 50 troopers to patrol the NFL Draft footprint daily, while the Wayne County Sheriff's Office will assign 70 deputies to the event.

The NFL revealed that Big Sean, Bazzi, the Detroit Youth Choir and more local artists will perform as part of the concert series for the 2024 NFL Draft in Detroit.

The No. 1 pick in this year's WNBA draft is "going to raise all boats" for players in the league, one expert said.

More than 20 people have been convicted in the case, many of them onetime NBA players.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.

Former Michigan Police Chief Tressa Beltran has pleaded guilty to drug charges, according to the Michigan Attorney General's office.

Two U.S. officials confirm to CBS News that an Israeli missile has hit Iran, in apparent retaliation for last weekend's drone and missile attack on the Jewish state.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

In response, one House Democrat said she is surprised to continue having these conversations.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

General Motors and real estate firm Bedrock announced Monday the automaker's headquarters will move to the Hudson's Detroit development in downtown Detroit from the Renaissance Center.

With U.S. electric vehicle sales starting to slow, Ford Motor Co. says it will delay rolling out new electric pickup trucks and a new large electric SUV as it adds gas-electric hybrids to its model lineup.

New vehicle sales in the U.S. rose nearly 5% from January through March, as buyers stayed in the market despite high interest rates.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Taylor Swift took to social media hours ahead of the expected release of her new album "The Tortured Poets Department."

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.

Charli XCX and Troye Sivan are teaming up to co-headline an arena tour across North America, which will kick off here in Detroit.

ABBA, Blondie and The Notorious B.I.G. are entering America's audio canon.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Tesla's stock price has tumbled 39% this year amid concerns about the electric vehicle maker's slowing growth.

Taylor Swift took to social media hours ahead of the expected release of her new album "The Tortured Poets Department."

Scientists are using a range of tools to protect the endangered wildlife that could disappear in coming decades.

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.

Health officials are warning consumers not to consume the Infinite Herbs basil sold at Trader Joe's after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

MDARD officials say the virus, which was first detected in Michigan in 2022, was found in Newaygo County, making it the seventh flock in the state in the last two years.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform. The unveiling comes hours after they were leaked on social media.

On the ground, the Michigan State Police will send at least 50 troopers to patrol the NFL Draft footprint daily, while the Wayne County Sheriff's Office will assign 70 deputies to the event.

The NFL revealed that Big Sean, Bazzi, the Detroit Youth Choir and more local artists will perform as part of the concert series for the 2024 NFL Draft in Detroit.

The No. 1 pick in this year's WNBA draft is "going to raise all boats" for players in the league, one expert said.

More than 20 people have been convicted in the case, many of them onetime NBA players.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

Riney, who has spent his 46-year career at Henry Ford Health, was named one of Modern Healthcare's 100 Most Influential People.

Iraqi Prime Minister Mohammed Shia al-Sudani traveled to Michigan on Thursday to meet with the state's large Iraqi community and update them on escalating tensions in the Middle East.

Detroit Lions unveiled new uniforms on Thursday, including a black alternate uniform.

In celebration of Arab American Heritage Month, CBS News Detroit takes a look into the culture in Metro Detroit, highlighting the history, leadership, food, and comedy.

Behind every athlete is a mother supporting them behind the scenes. On April 21, the "My Mom's First Fan" event celebrates Metro Detroit football mothers. Khary Frazier, founder of Detroit is Different, and Constance "Maspice" Davis, the mother of retired NFL player Anthony "Spice" Adams, joined CBS News Detroit to talk about the upcoming event.

Rain overnight will break early tomorrow morning while cooler air rushes in to kickstart our weekend.