
NFL Draft in Detroit. What to know about registration, security, road closures and more
Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.
Watch CBS News

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Coach Reggie "Boobie" Wynns is the founder of Rising Stars, a supplemental football program dedicated to making young athletes better players while preparing them for the rigorous world of college sports recruiting.

Marshella Chidester, 66, is suspected to have been driving drunk when she allegedly crashed into a building at the Swan Boat Club property, killing two children.

After a set of hate crime bills didn't make it across the finish line to Gov. Gretchen Whitmer's desk in 2023, lawmakers made changes and are seeing new movement.

The USDA had floated banning flavored milk options from some school lunches.
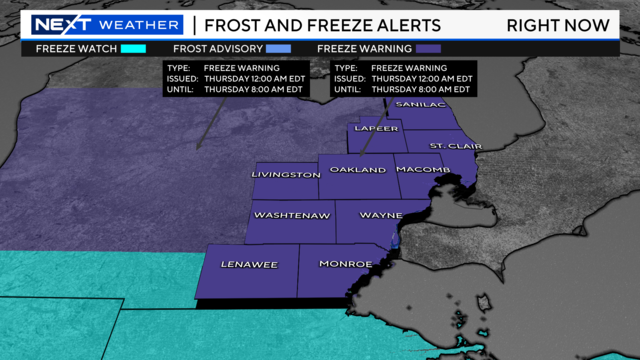
There is an updated freeze warning for all counties in Southeast Michigan as temperatures drop into the 20s Wednesday night through tomorrow morning.

Two magnet schools in Metro Detroit have ranked among the top 20 best high schools in the country.

Crews will complete repairs on a sinkhole in Oakland County after a 12-inch collapsed sewer.





A Metro Detroit football coach is pushing past his pain to pursue his purpose of helping others.

After a set of hate crime bills didn't make it across the finish line to Gov. Gretchen Whitmer's desk in 2023, lawmakers made changes and are seeing new movement.
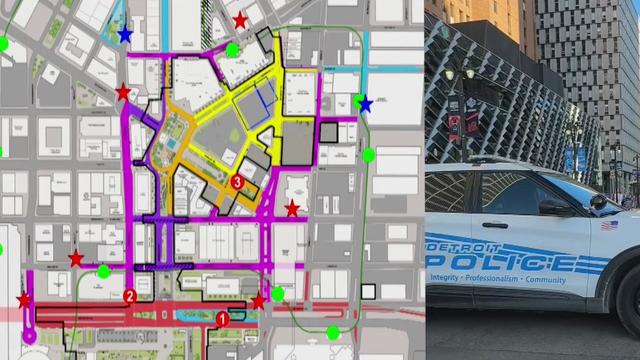
The fifth and final phase of road closures for the 2024 NFL Draft started midweek.

Jeanette Schneider, president of RE/MAX of Southeastern Michigan, shares more on how single women are outpacing single men as homebuyers.

If you're headed to the 2024 NFL Draft this week in Detroit, we've got you covered with transportation options.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

Coach Reggie "Boobie" Wynns is the founder of Rising Stars, a supplemental football program dedicated to making young athletes better players while preparing them for the rigorous world of college sports recruiting.

The NFL draft has taken the show on the road for a decade, giving cities around the country a chance to be in the spotlight.

Employees of the company tasked with rapidly personalizing jerseys for each first-round NFL draft pick as they are announced don't need to travel very far for this year's player selections in Detroit.

Over the next 72 hours, the NFL is unpacking and installing all the equipment fans will enjoy when the gates open at noon on Thursday..

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Coach Reggie "Boobie" Wynns is the founder of Rising Stars, a supplemental football program dedicated to making young athletes better players while preparing them for the rigorous world of college sports recruiting.

Marshella Chidester, 66, is suspected to have been driving drunk when she allegedly crashed into a building at the Swan Boat Club property, killing two children.

After a set of hate crime bills didn't make it across the finish line to Gov. Gretchen Whitmer's desk in 2023, lawmakers made changes and are seeing new movement.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Despite a small dip in U.S. vehicle sales, General Motors' first-quarter net income rose more than 25% on strong deliveries of pickup trucks and other higher-profit vehicles.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

General Motors and real estate firm Bedrock announced Monday the automaker's headquarters will move to the Hudson's Detroit development in downtown Detroit from the Renaissance Center.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

"Ultimately we think this is a better approach that reflects the evolution of the business," Netflix Co-CEO Greg Peters said on an earnings call.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

Guitar legend Dickey Betts, who co-founded the Allman Brothers Band and wrote their biggest hit, "Ramblin' Man," has died.

Expanded federal overtime rule could result in employers paying workers an additional $1.5 billion, according to one estimate.

There are no cameras allowed in the court where Trump is being tried on 34 felony counts stemming from a "hush money" payment before the 2016 election.

Travelers often spend more than they need to for airfare, experts say. Here's what to know about paying for add-ons like your seat assignment.

Jurors in former President Donald Trump's trial in New York heard testimony from a former media executive about his efforts to bury negative stories about Trump before the 2016 presidential election.

Ryan Watson could face at least 12 years in prison in Turks and Caicos after airport security allegedly found four rounds of hunting ammo in his carry-on.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.
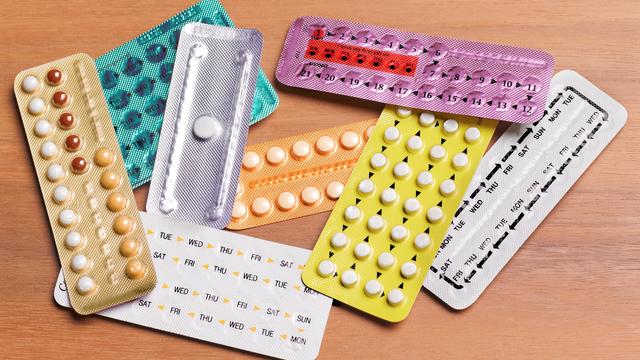
The Michigan Legislature is considering two bills that could make getting access to hormonal birth control a little easier.

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

MDARD officials say the virus, which was first detected in Michigan in 2022, was found in Newaygo County, making it the seventh flock in the state in the last two years.

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

Coach Reggie "Boobie" Wynns is the founder of Rising Stars, a supplemental football program dedicated to making young athletes better players while preparing them for the rigorous world of college sports recruiting.

The NFL draft has taken the show on the road for a decade, giving cities around the country a chance to be in the spotlight.

Employees of the company tasked with rapidly personalizing jerseys for each first-round NFL draft pick as they are announced don't need to travel very far for this year's player selections in Detroit.

Over the next 72 hours, the NFL is unpacking and installing all the equipment fans will enjoy when the gates open at noon on Thursday..
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

A Metro Detroit football coach is pushing past his pain to pursue his purpose of helping others.

After a set of hate crime bills didn't make it across the finish line to Gov. Gretchen Whitmer's desk in 2023, lawmakers made changes and are seeing new movement.

The fifth and final phase of road closures for the 2024 NFL Draft started midweek.

Jeanette Schneider, president of RE/MAX of Southeastern Michigan, shares more on how single women are outpacing single men as homebuyers.

If you're headed to the 2024 NFL Draft this week in Detroit, we've got you covered with transportation options.