
Man charged in deadly Michigan explosion was trying to flee the country, police report
Noor Kestou was taken into custody by Macomb County detectives on Wednesday, April 24.
Watch CBS News

Noor Kestou was taken into custody by Macomb County detectives on Wednesday, April 24.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

The Detroit Lions made a move to address their secondary, grabbing Alabama cornerback Terrion Arnold with the No. 24 pick in the NFL draft after a trade with Dallas.

One of the biggest events in the NFL year kicked off Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

The woman charged in a deadly crash at a Monroe County birthday party that killed two children has posted bond two days later, according to the sheriff's office.

The driver was arrested after police said she had a blood alcohol content of .17%.

Here's everything to know about registration, security policies, road closures, concerts and more for the NFL Draft.

A new rule will affect frozen breaded and stuffed raw chicken products that appear to be fully cooked but are only heat-treated.





Sunshine this afternoon, rain returns late. NEXT Weather Meteorologist Kylee Miller has your weekend forecast.

Former Detroit Lions defensive tackle Andre Fluellen is giving back to Detroit students in May by hosting 40 students for a dinner at Prime + Proper.
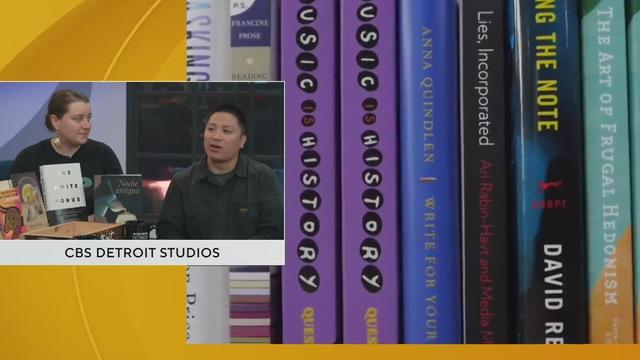
We're celebrating National Independent Bookstore Day with 27th Letter Books in Detroit.

CBS News Detroit's Jordan Burrows takes us on a tour of the food and drink options at the 2024 NFL Draft in Detroit.

A Commerce man facing manslaughter charges in connection with a deadly, massive explosion at a Clinton Township business in March had a one-way ticket to Hong Kong before being arrested, police say.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

NFL Commissioner Roger Goodell announced that more than 275,000 fans attended the first day on Thursday, according to a spokesperson.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

The Detroit Lions made a move to address their secondary, grabbing Alabama cornerback Terrion Arnold with the No. 24 pick in the NFL draft after a trade with Dallas.

The Detroit Lions selected Alabama cornerback Terrion Arnold on Thursday in the first round of the 2024 NFL Draft.

Michigan football quarterback J.J. McCarthy is starting his NFL career with the Minnesota Vikings after being selected No. 10 in the first round of the 2024 NFL Draft.

An unprecedented six of the first 12 picks were quarterbacks, an NFL Draft record.

The NFL draft gives players a chance to show their style and many take advantage with custom-made suits.

Noor Kestou was taken into custody by Macomb County detectives on Wednesday, April 24.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

The Detroit Lions made a move to address their secondary, grabbing Alabama cornerback Terrion Arnold with the No. 24 pick in the NFL draft after a trade with Dallas.

One of the biggest events in the NFL year kicked off Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Despite a small dip in U.S. vehicle sales, General Motors' first-quarter net income rose more than 25% on strong deliveries of pickup trucks and other higher-profit vehicles.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

NFL Commissioner Roger Goodell announced that more than 275,000 fans attended the first day on Thursday, according to a spokesperson.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

A new rule will affect frozen breaded and stuffed raw chicken products that appear to be fully cooked but are only heat-treated.

It's been a decade since the Flint water crisis began. Residents told CBS News the scandal still weighs heavily on the city.

An unprecedented six of the first 12 picks were quarterbacks, an NFL Draft record.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.
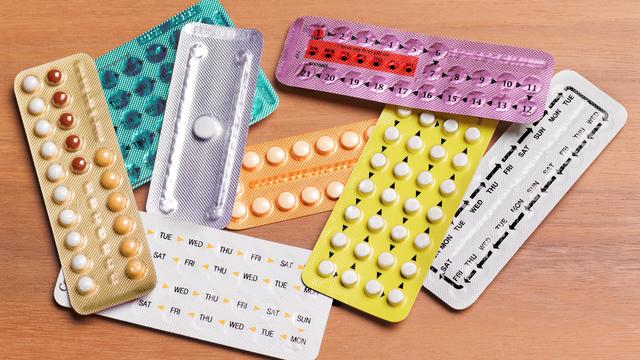
The Michigan Legislature is considering two bills that could make getting access to hormonal birth control a little easier.

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

The Detroit Lions made a move to address their secondary, grabbing Alabama cornerback Terrion Arnold with the No. 24 pick in the NFL draft after a trade with Dallas.

The Detroit Lions selected Alabama cornerback Terrion Arnold on Thursday in the first round of the 2024 NFL Draft.

Michigan football quarterback J.J. McCarthy is starting his NFL career with the Minnesota Vikings after being selected No. 10 in the first round of the 2024 NFL Draft.

An unprecedented six of the first 12 picks were quarterbacks, an NFL Draft record.

The NFL draft gives players a chance to show their style and many take advantage with custom-made suits.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

The issue of abortion and IVF was also discussed during the roundtable, along with thoughts on how they might sway voters this fall.

Sunshine this afternoon, rain returns late. NEXT Weather Meteorologist Kylee Miller has your weekend forecast.

Former Detroit Lions defensive tackle Andre Fluellen is giving back to Detroit students in May by hosting 40 students for a dinner at Prime + Proper.

We're celebrating National Independent Bookstore Day with 27th Letter Books in Detroit.

CBS News Detroit's Jordan Burrows takes us on a tour of the food and drink options at the 2024 NFL Draft in Detroit.

A Commerce man facing manslaughter charges in connection with a deadly, massive explosion at a Clinton Township business in March had a one-way ticket to Hong Kong before being arrested, police say.