
Trader Joe's pulls fresh basil in 29 states after salmonella outbreak
Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.
Watch CBS News

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

After traces of lead in applesauce poisons nearly 470 people, mostly young kids, FDA finds the metal in more food items.

Connecticut-based company Valisure issued a citizen petition to the FDA saying it found "high levels of benzene" in acne products containing benzoyl peroxide, including Proactiv and Clearasil.

In January, the FDA changed regulations to allow retail pharmacies to sell the drug mifepristone.

Multiple brands sold by Walmart and CVS recalled as FDA cites unsterile conditions at Indian manufacturing plant.
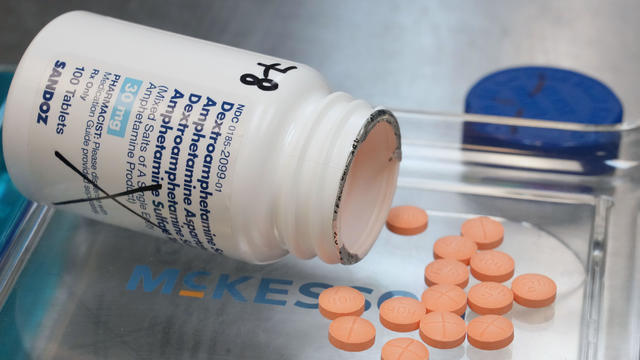
Generic versions of the ADHD medications Adderall, Vyvanse, and Concerta remain in shortage in 2024.

Ecuador's authorities allege that Carlos Aguilera, a cinnamon grinder in that country, is "the likely source" of the contaminated spices that led to the recall of WanaBana applesauce.

A U.S. Department of Agriculture grant is boosting schools' opportunities to make sure students are introduced to good but healthy foods.
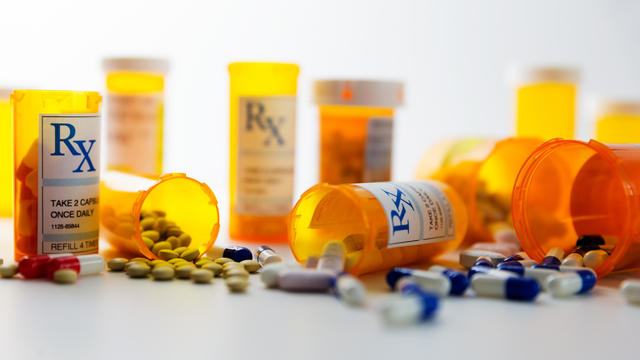
Azurity Pharmaceuticals is recalling Zanzedi after a pharmacist found a pill with sedative effects in a package of the stimulant drug.

FDA testing found "extremely high levels of lead contamination" in the cinnamon applesauce pouches, which have been recalled.

Quaker Oats expands prior recall to include more granola bars, cereals and a snack mix possibly tainted with bacteria.

Five flavors of Benny T's Vesta dry hot sauce are being recalled for having labels that fail to disclose allergens in the products.

Among the supermarket foods tested, Annie's Organic Cheesy Ravioli, Del Monte sliced peaches and Chicken of the Sea pink salmon, had the most phthalates per nanogram.

Ozempic and other GLP-1 RA drugs, including Wegovy, Mounjaro and Zepbound, are being scrutinized.
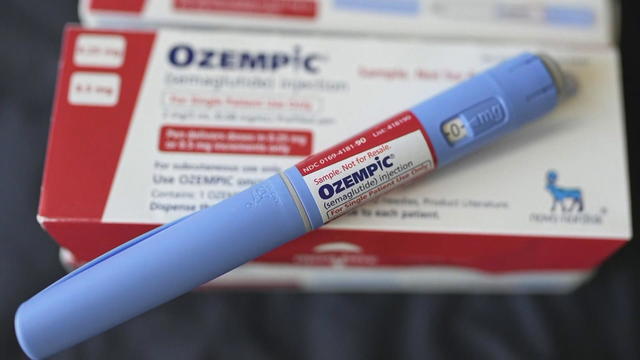
The FDA and drugmaker Novo Nordisk are working to identify thousands of counterfeit Ozempic drugs seized in an ongoing investigation.

Coca-Cola's recall impacts 12-pack cans of soda distributed in Alabama, Mississippi and Florida, the FDA said in a filing.

Consuming some caffeine is typically harmless for adults, but having too much can be dangerous. Here's what to know about potential health effects.

The FDA and the manufacturer were alerted to Profemur titanium hips breaking inside U.S. patients as of 2005. It took 15 years to recall the devices. Many fractures could have been avoided.

The illegal Neptune's Fix brand supplements claim to contain tianeptine, known as "gas station heroin."

CDC investigating fruit from HMC Farms after one person died and 10 are hospitalized following a listeria outbreak.

It is unclear if the counterfeit drugs, which many people seek for weight loss, caused the hospitalizations.

The Food and Drug Administration said they are looking into the death of a student with a heart condition whose parents allege died after consuming a caffeinated drink at Panera Bread.

CVS won't sell decongestants that contain phenylephrine as the only active ingredient after an FDA panel called the ingredient ineffective.
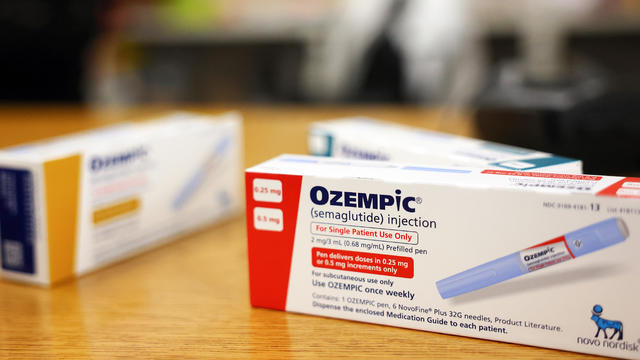
The FDA stopped short of saying the potentially life-threatening condition was caused by the drugs, which have become popular for weight loss.

ASR Pharma said it was surprised and "deeply disappointed" by the FDA request for more trial data on its Neffy nasal spray for severe allergic reactions.

West Michigan University defensive end Marshawn Kneeland is going to Texas and being selected by the Dallas Cowboys in the second round of the 2024 NFL Draft.

The Detroit Lions have selected another cornerback in the 2024 NFL Draft! This time, they've picked Ennis Rakestraw Jr. from Missouri.

Michigan defensive tackle Kris Jenkins and cornerback Mike Sainristil are the next Wolverines to be selected in the 2024 NFL Draft.

Detroit is getting close to setting the record for the number of fans attending the NFL Draft and it'll just take about 100,000 more people to do it.
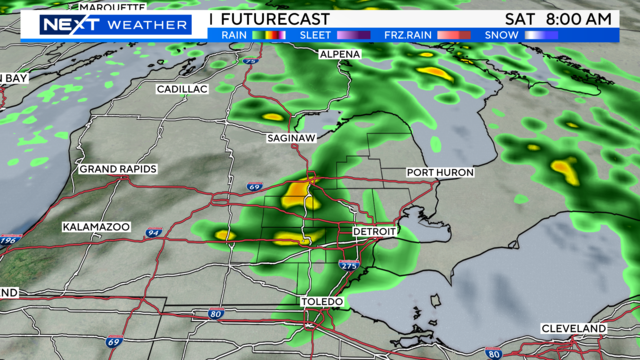
Expect high temperatures in the mid-to-upper 70s for the weekend, with some wind and rain.

West Michigan University defensive end Marshawn Kneeland is going to Texas and being selected by the Dallas Cowboys in the second round of the 2024 NFL Draft.

Noor Kestou was taken into custody by Macomb County detectives on Wednesday, April 24.

One of the biggest events in the NFL year kicked off Thursday in downtown Detroit. Aside from community events happening in the area, many people are here to see who the teams will select in the 2024 NFL Draft.

The Detroit Lions have selected another cornerback in the 2024 NFL Draft! This time, they've picked Ennis Rakestraw Jr. from Missouri.

Detroit City FC heads on the road this weekend to face off against Eastern Conference rival Pittsburgh Riverhounds SC.

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

Jeep maker Stellantis is planning to lay off an unspecified number of workers at its U.S. factories in the coming months to deal with a rapidly changing global auto market, the company said Tuesday.

Despite a small dip in U.S. vehicle sales, General Motors' first-quarter net income rose more than 25% on strong deliveries of pickup trucks and other higher-profit vehicles.

Workers at Mercedes-Benz factories near Tuscaloosa, Alabama, will vote next month on whether they want to be represented by the United Auto Workers union.

After leading the United Auto Workers on its historic strike against the Detroit Three automakers, Shawn Fain has been named on Time's list of the most influential people of 2024.

NFL Commissioner Roger Goodell announced that more than 275,000 fans attended the first day on Thursday, according to a spokesperson.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Many of the events taking place at the Corner Ballpark during the three-day event will be for kids, but go down Michigan Avenue, and there will be plenty for adults to do.

From art and architecture to entertainment, the Motor City is at the centerpiece of the draft.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

With a relatively low average monthly cost of living and a low crime rate, this little-known town has a lot to offer retirees according to one report.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

A new rule will affect frozen breaded and stuffed raw chicken products that appear to be fully cooked but are only heat-treated.

It's been a decade since the Flint water crisis began. Residents told CBS News the scandal still weighs heavily on the city.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.
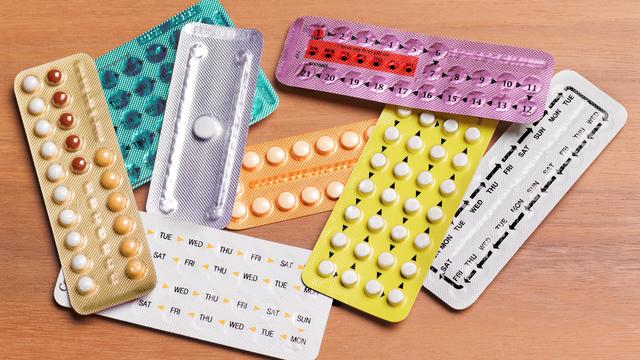
The Michigan Legislature is considering two bills that could make getting access to hormonal birth control a little easier.

Health officials are warning consumers not to consume Infinite Herbs basil sold at some Trader Joe's and Dierberg's stores after 12 people were sickened.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

West Michigan University defensive end Marshawn Kneeland is going to Texas and being selected by the Dallas Cowboys in the second round of the 2024 NFL Draft.

The Detroit Lions have selected another cornerback in the 2024 NFL Draft! This time, they've picked Ennis Rakestraw Jr. from Missouri.

Michigan defensive tackle Kris Jenkins and cornerback Mike Sainristil are the next Wolverines to be selected in the 2024 NFL Draft.

Detroit City FC heads on the road this weekend to face off against Eastern Conference rival Pittsburgh Riverhounds SC.

The National Bobblehead Hall of Fame and Museum on Friday unveiled the new limited edition bobblehead featuring Oakland University basketball player Jack Golhke.
By the time this presidential race is over, Biden, Trump and their entourages may not need Michigan driver's licenses, but they'll know their way around and where the orange barrel construction zones are enough to void them on their way to campaign events.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Three leaders talked about how the region is doing to help people deal with mental health issues.

It's Day 2 of the 2024 NFL Draft, and once again, Detroit Lions fans are showing their pride in the Motor City. We caught up with a few fans to talk about their experience at the draft.

The Detroit Lions made a move to address their secondary, grabbing Alabama cornerback Terrion Arnold with the No. 24 pick in the NFL draft after a trade with Dallas on Thursday night.

A Commerce man facing manslaughter charges in connection with a deadly, massive explosion at a Clinton Township business in March had a one-way ticket to Hong Kong before being arrested, police say.

Jeff Lesson of WWJ Newsradio 950 and 97.1 The Ticket shares his predictions for the second and third rounds of the 2024 NFL Draft.

After a record-setting first-round attendance, Detroit Police Chief James White discusses security at the 2024 NFL Draft in Detroit.