
Michigan program will allow incarcerated mothers to send breastmilk to infants
Inmates at Michigan's Huron Valley Correctional Facility will be allowed to send their breastmilk home for their children under a new program.
Watch CBS News

Inmates at Michigan's Huron Valley Correctional Facility will be allowed to send their breastmilk home for their children under a new program.

A restaurant in Detroit was named the most beautiful in Michigan, according to a list published in People Magazine.

Zoo officials announced that the lion is moving to the Great Plains Zoo in Sioux Falls, South Dakota, after more than a decade in Michigan.

Police in Michigan say a startling discovery was made on the roof of a Michigan grocery store: A woman was living inside the store sign for roughly a year.

A neighborhood-based nonprofit housing corporation is making a lot of progress with constructing its affordable housing complex at Orchard Street and Lahser Road in Detroit's Old Redford neighborhood.

Weather officials say the tornado reached an estimated peak wind of 135 mph and touched down near the intersection of S. 10th Street and W. R Avenue. It traveled for about 11 miles, damaging home roofs and sidings.

The bill stalled earlier this week after senators from Virginia and Maryland objected to a provision that would allow an additional 10 flights a day to and from Ronald Reagan Washington National Airport.

Apple's "Crush!" advertisement for the new iPad Pro features a myriad of artistic tools getting smashed in a large hydraulic press.

Police are looking for a man who allegedly shouted antisemitic remarks toward two women last week outside of a synagogue in Oak Park.

Police are looking for a man who allegedly shouted antisemitic remarks toward two women last week outside of a synagogue in Oak Park.

Inmates at Michigan's Huron Valley Correctional Facility will be allowed to send their breastmilk home for their children under a new program.

A neighborhood-based nonprofit housing corporation is making a lot of progress with constructing its affordable housing complex at Orchard Street and Lahser Road in Detroit's Old Redford neighborhood.

Graduation season is underway, and one thing many will think about is the food for the celebration. Dewayne Gordon and Eric Fuller with Gus's World Famous Fried Chicken provide tips on catering.

For many moms, motherhood can have an impact on their mental health. Dr. Charletta Dennis, medical director at Priority Health, joined CBS News Detroit to break down mental health at every stage of motherhood.

The roundtable discusses mobility and the importance of marque events like the 2024 NFL Draft, the Detroit Auto Show and more.
By the time this presidential race is over, Biden, Trump and their entourages may not need Michigan driver's licenses, but they'll know their way around and where the orange barrel construction zones are enough to void them on their way to campaign events.

This will be the first baby for Hailey and Justin Beiber, who announced their pregnancy after more than five years of marriage.

UmaSofia Srivastava resigned as Miss Teen USA on Wednesday, just two days after Noelia Voigt stepped down as Miss USA.

Brian Fox, a fellow producer and engineer at Albini's Electrical Audio studio in Chicago, confirmed Albini passed away Tuesday night from a heart attack.

The weather was perfect for the regional Special Olympics held at Macomb County Community College in Warren.

The Olympic flame arrived in France aboard a 19th century tall ship to kick off a 7,500-mile journey to the Paris Summer Games.

Detroit will officially debut its uniforms at Comerica Park on Friday against the Houston Astros.

Here are the different ticket options fans can now purchase for the 2024 Rocket Mortgage Classic.

Tom Brady took his share of barbs from comedians, former teammates and his longtime coach during a made-for-streaming comedy live event on Netflix -- but one joke seemed to anger him.

Inmates at Michigan's Huron Valley Correctional Facility will be allowed to send their breastmilk home for their children under a new program.

A restaurant in Detroit was named the most beautiful in Michigan, according to a list published in People Magazine.

Zoo officials announced that the lion is moving to the Great Plains Zoo in Sioux Falls, South Dakota, after more than a decade in Michigan.

Police in Michigan say a startling discovery was made on the roof of a Michigan grocery store: A woman was living inside the store sign for roughly a year.

A neighborhood-based nonprofit housing corporation is making a lot of progress with constructing its affordable housing complex at Orchard Street and Lahser Road in Detroit's Old Redford neighborhood.

United Auto Workers union members at Stellantis' Warren Stamping Plant voted on Monday to authorize a strike over health and safety concerns.

Members of UAW Local 869 are scheduled to vote on May 6 over safety concerns at the plant.

Technology originally developed for homeland security is now available in Michigan to inspect every inch of your car. Developers say UVeye is like getting an MRI for your vehicle.

A bill introduced to the Michigan Legislature is looking to decrease noise pollution around Metro Detroit, more particularly along Woodward Avenue.

Ford Motor Co.'s first-quarter net income fell 24% from a year ago as the company's combustion engine vehicle unit saw revenue and sales decline.

This will be the first baby for Hailey and Justin Beiber, who announced their pregnancy after more than five years of marriage.

UmaSofia Srivastava resigned as Miss Teen USA on Wednesday, just two days after Noelia Voigt stepped down as Miss USA.

Brian Fox, a fellow producer and engineer at Albini's Electrical Audio studio in Chicago, confirmed Albini passed away Tuesday night from a heart attack.

'Bob Hearts Abishola', the acclaimed comedy, is signing off after its fifth season on CBS.

Tom Brady took his share of barbs from comedians, former teammates and his longtime coach during a made-for-streaming comedy live event on Netflix -- but one joke seemed to anger him.

The bill stalled earlier this week after senators from Virginia and Maryland objected to a provision that would allow an additional 10 flights a day to and from Ronald Reagan Washington National Airport.

Apple's "Crush!" advertisement for the new iPad Pro features a myriad of artistic tools getting smashed in a large hydraulic press.

The fitness chain's $10 monthly membership is one of few things that had remained unchanged since 1998 — until now.

At its height, the Chevy Malibu won Motor Trend Car of the Year 1997 because of its smooth ride and fuel economy.

This will be the first baby for Hailey and Justin Beiber, who announced their pregnancy after more than five years of marriage.

The fitness chain's $10 monthly membership is one of few things that had remained unchanged since 1998 — until now.

Elon Musk's Neuralink finds a brain-computer interface device captured less data a month after implant surgery.
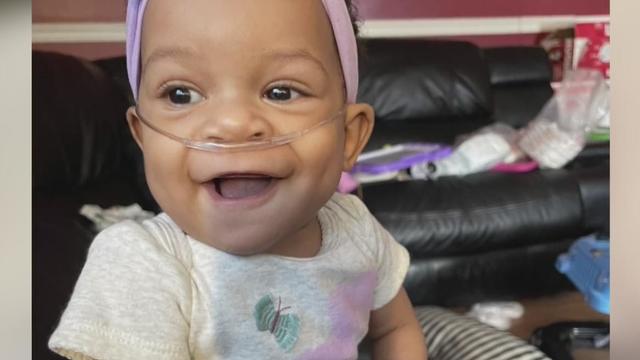
A Metro Detroit mother wants her 1-year-old transferred to a hospital out of state after she says she raised concerns about her daughter's care at Children's Hospital of Michigan.

An 11-year-old boy in Warren has made medical history. He is one of two children in the world to receive an experimental gene therapy procedure to restore vision.

Panera is phasing out a highly caffeinated selection of lemonade beverages that's at the center of several lawsuits.

The weather was perfect for the regional Special Olympics held at Macomb County Community College in Warren.

The Olympic flame arrived in France aboard a 19th century tall ship to kick off a 7,500-mile journey to the Paris Summer Games.

Detroit will officially debut its uniforms at Comerica Park on Friday against the Houston Astros.

Here are the different ticket options fans can now purchase for the 2024 Rocket Mortgage Classic.

Tom Brady took his share of barbs from comedians, former teammates and his longtime coach during a made-for-streaming comedy live event on Netflix -- but one joke seemed to anger him.

The roundtable discusses mobility and the importance of marque events like the 2024 NFL Draft, the Detroit Auto Show and more.

By the time this presidential race is over, Biden, Trump and their entourages may not need Michigan driver's licenses, but they'll know their way around and where the orange barrel construction zones are enough to void them on their way to campaign events.
The Motor City is putting the final touches in place for the 2024 NFL Draft as it rolls into Detroit next week.

Michael Bickers, Regional President of PNC Southeast Michigan/Detroit, and Eric Poe, CEO of CURE Auto Insurance, appeared on CBS Detroit's Michigan Matters.

Businesses across Michigan are feeling optimistic about the future after weathering the historic 100-year health pandemic that impacted every company in one way or another.

Police are looking for a man who allegedly shouted antisemitic remarks toward two women last week outside of a synagogue in Oak Park.

Inmates at Michigan's Huron Valley Correctional Facility will be allowed to send their breastmilk home for their children under a new program.

A neighborhood-based nonprofit housing corporation is making a lot of progress with constructing its affordable housing complex at Orchard Street and Lahser Road in Detroit's Old Redford neighborhood.

Graduation season is underway, and one thing many will think about is the food for the celebration. Dewayne Gordon and Eric Fuller with Gus's World Famous Fried Chicken provide tips on catering.

For many moms, motherhood can have an impact on their mental health. Dr. Charletta Dennis, medical director at Priority Health, joined CBS News Detroit to break down mental health at every stage of motherhood.